About the TROPIC Database
TROPIC (Thermodynamics of Ring-Opening Polymerisation Informatics Collection) is an open-source database of experimental and computational thermodynamic parameters for ring-opening polymerization (ROP) reactions. Our goal is to facilitate the data-driven discovery of novel, functional polymers that are primed for chemical recycling.
Data Collection & Curation
The data in this database has been manually collected from peer-reviewed academic literature. Each entry is linked to its source via a Digital Object Identifier (DOI), allowing for easy access to the original publication and its metadata.
We include key experimental conditions, such as the reaction medium and monomer concentration, as these factors significantly influence thermodynamic values. To ensure data quality, we use a flagging system to highlight entries that are incomplete or appear inconsistent.
Molecule Classes
The current dataset focuses on several classes of polar cyclic monomers (PCMs) used in ring-opening polymerizations.
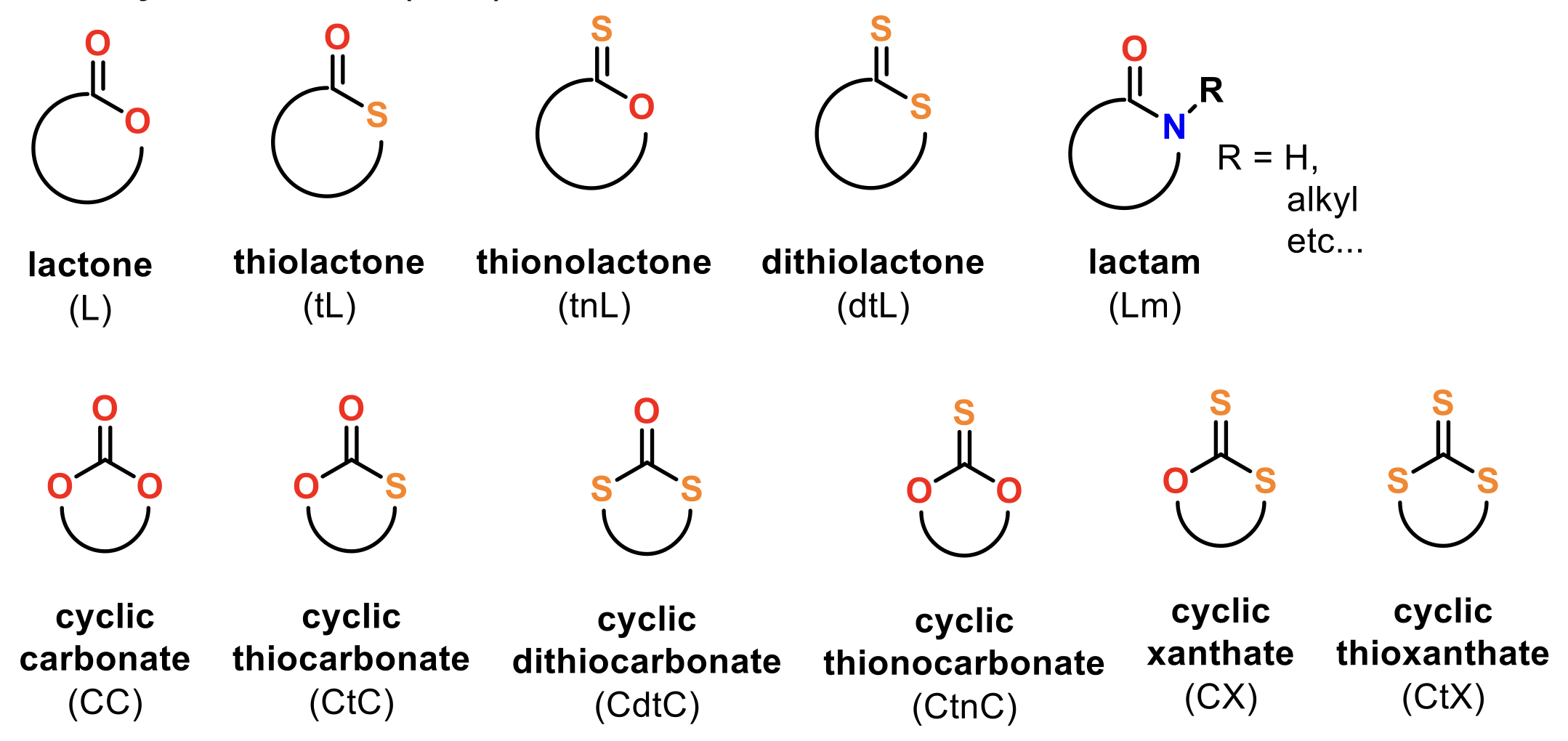
| Abbreviation | Monomer Class |
|---|---|
| L | Lactone |
| CC | Cyclic Carbonate |
| Lm | Lactam |
| tL, tnL, dtL | Sulfur-containing Lactone Derivatives |
| CtC, CdtC, etc. | Sulfur-containing Cyclic Carbonate Derivatives |
Reaction Types
The database classifies reactions into four distinct categories to provide clear context for the thermodynamic data.
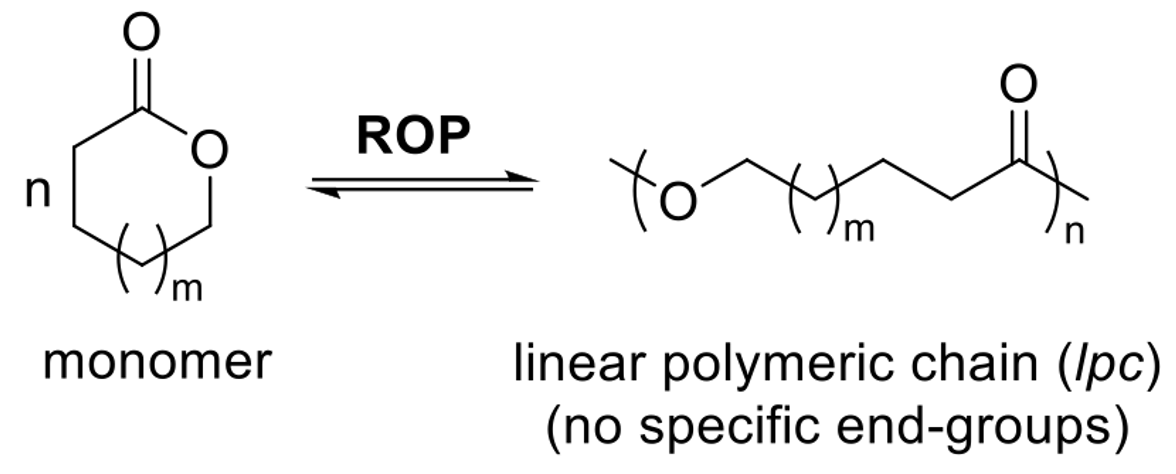
ROP (Ring-Opening Polymerization)
An experimental reaction where cyclic monomers form long-chain linear polymers.
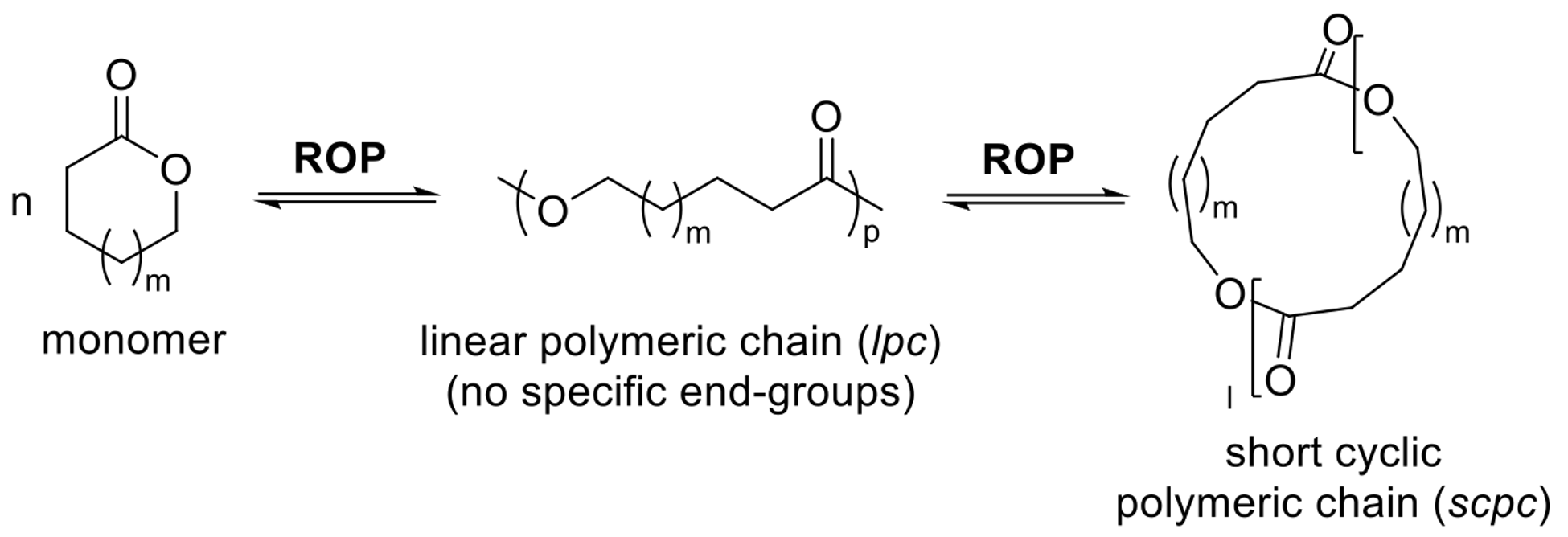
RCE (Ring-Chain Equilibrium)
An experimental equilibrium between all ring sizes (including monomer) and the final polymer.
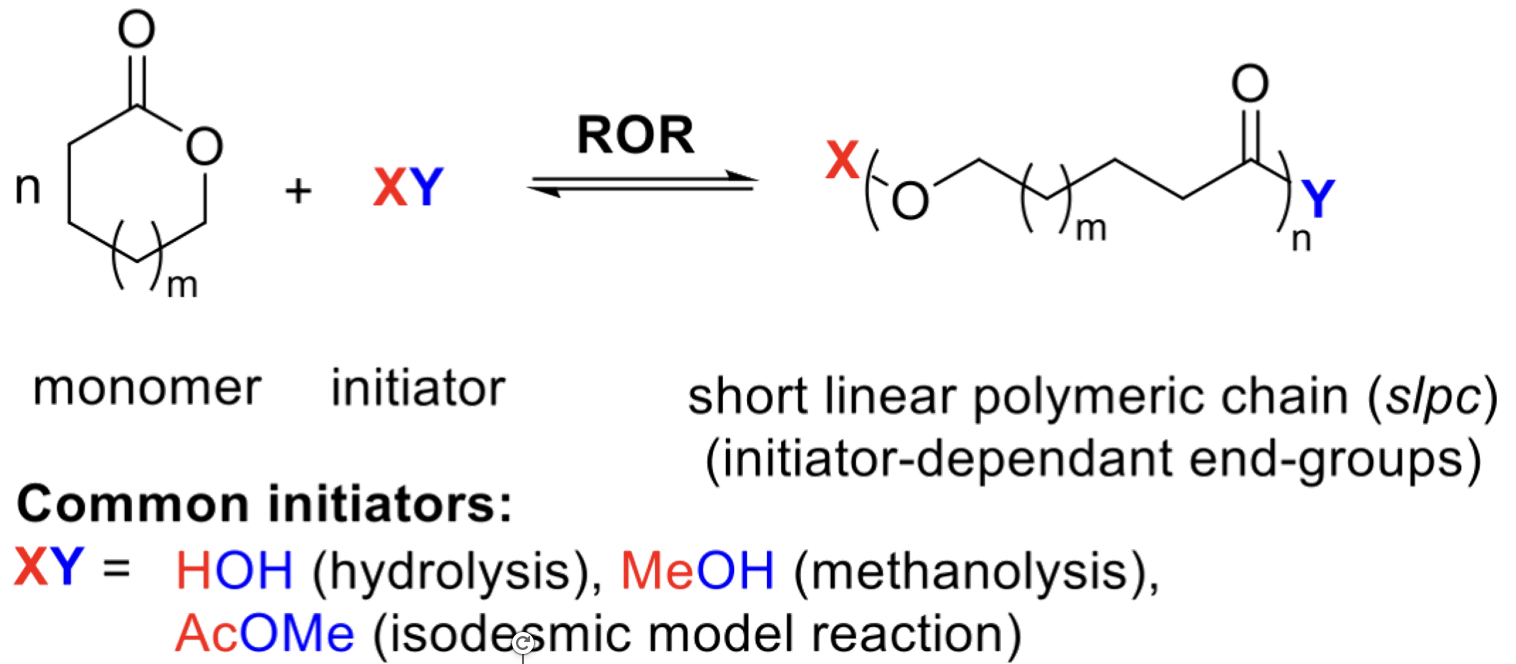
ROR (Ring-Opening Reaction)
A model reaction, computational or experimental, where one or a few monomers are opened to form a short linear chain.
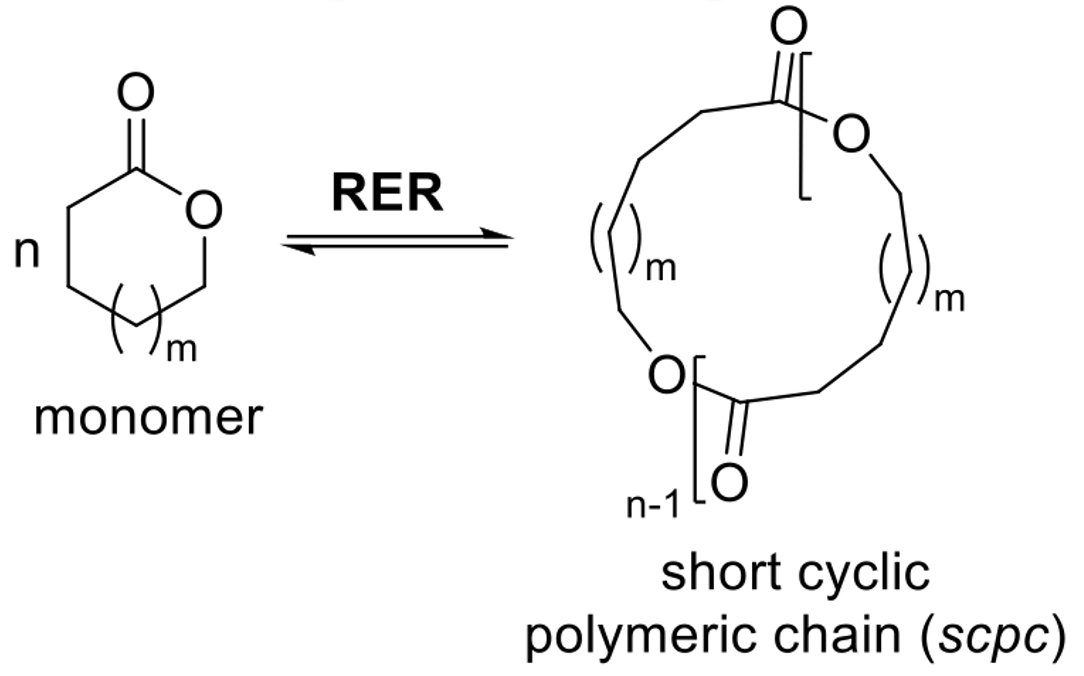
RER (Ring-Expansion Reaction)
A computational model where cyclic monomers form short cyclic polymer chains or "loops."
Thermodynamic Principles
The ceiling temperature () is a critical parameter for chemical recyclability. It represents the temperature at which polymerization and depolymerization are in equilibrium. It is derived from the enthalpy () and entropy () of polymerization.
The relationship is defined by the Dainton equation, where is the monomer concentration at equilibrium:
Methodologies
Experimental Methods
Van't Hoff Analysis
Determining and from the monomer equilibrium concentration at various temperatures, often measured via NMR spectroscopy.
Differential Scanning Calorimetry (DSC)
A thermal analysis technique used to measure heat flow associated with polymer transitions.
Calorimetry
Direct measurement of heat changes during polymerization.
NMR Spectroscopy
Used to monitor monomer conversion and determine equilibrium concentrations.
Computational Methods
Density Functional Theory (DFT)
Used for Ring-Opening Reaction (ROR) models to calculate thermodynamic parameters. The specific functional andbasis_set are recorded.
Molecular Dynamics (MD)
Used to identify stable conformers before DFT calculations. Methods include classical (ffmd), ab initio (aimd), and machine-learned (mlmd) dynamics.
Semi-Empirical Methods
Quantum mechanical methods like xTB that offer a balance between speed and accuracy.